
Efficacy of NATPARin chronic hypoparathyroidism
In the multinational, phase III REPLACE study, NATPAR maintained serum calcium while reducing oral calcium and active vitamin D needs1,2
REPLACE study design (N=124)1
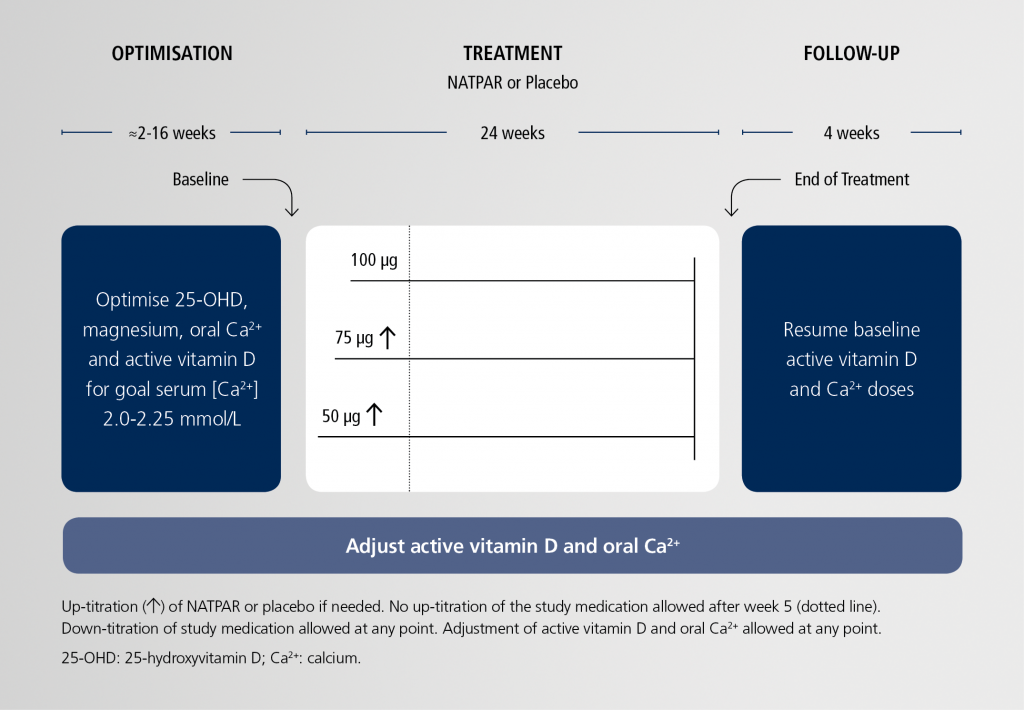
Primary and secondary endpoints
For the primary efficacy endpoint, patients had to fulfil all three components of a three-part response criterion at week 24:1,2
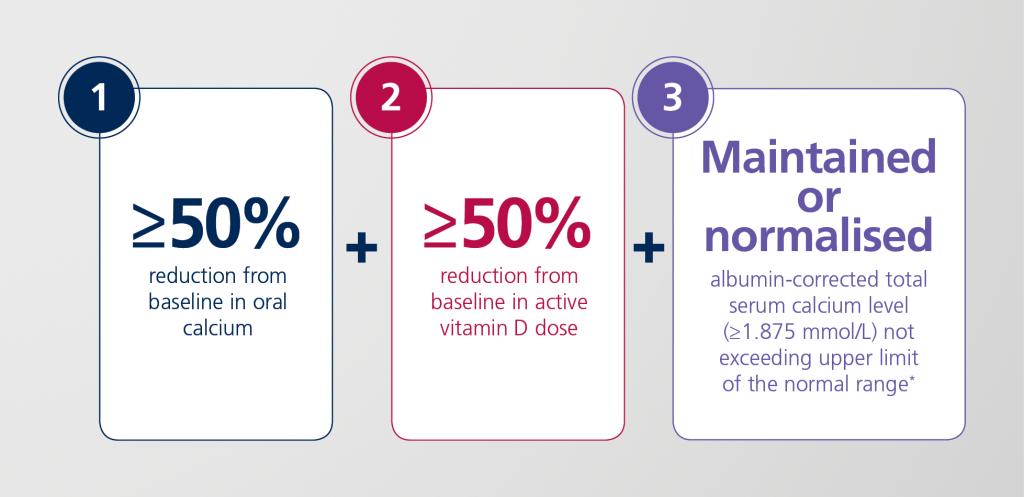
*Ideally between the target range of 2.0-2.25 mmol/L
Secondary endpoints included:2
- proportion of patients who achieved independence from active vitamin D while taking 500 mg per day or less of oral calcium at week 24
- percentage change from baseline in prescribed oral calcium dose by week 24
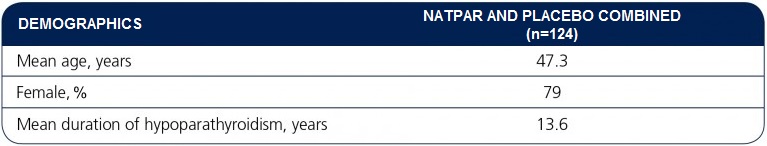
Patient demographics and baseline characteristics
1
NATPAR maintained serum calcium while reducing oral calcium and active vitamin D needs1,2
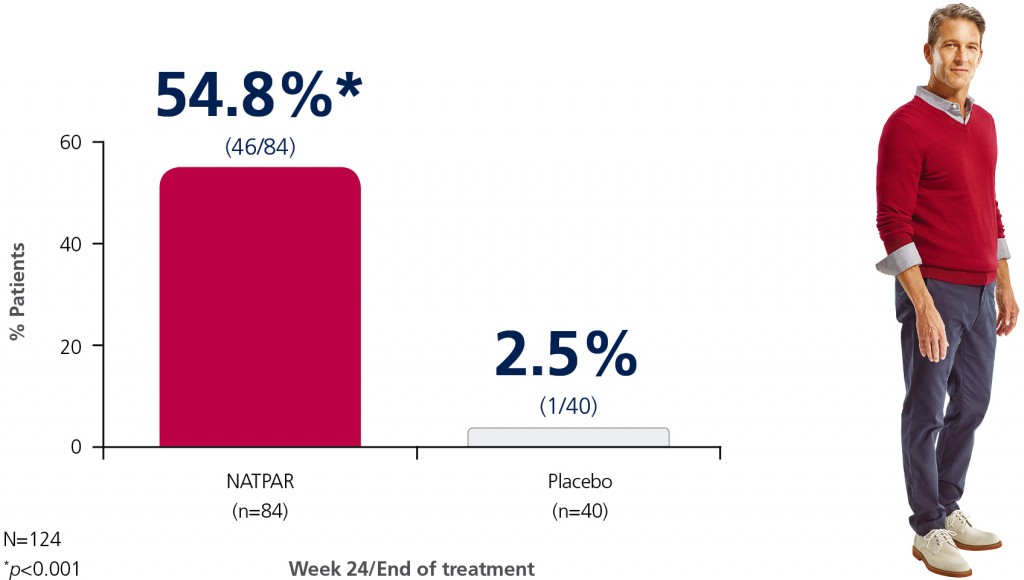
Approximately 55% of NATPAR patients maintained serum calcium at lower doses of calcium and vitamin D.1
Patients achieving the primary efficacy endpoint1
NATPAR helped 43% of patients eliminate active vitamin D and reduce oral calcium to 500 mg or less per day1
At week 24, for patients who completed the study, 43.0% of NATPAR-treated patients were independent of active vitamin D treatment and were receiving no more than 500 mg of calcium citrate, compared with 6.1% of placebo-treated patients (p<0.001)1

NATPAR demonstrated approximately 52% reduction in oral calcium1
The mean percentage change from baseline in oral calcium was -51.8% in patients receiving NATPAR compared with 6.5% in the placebo group (p<0.001).1

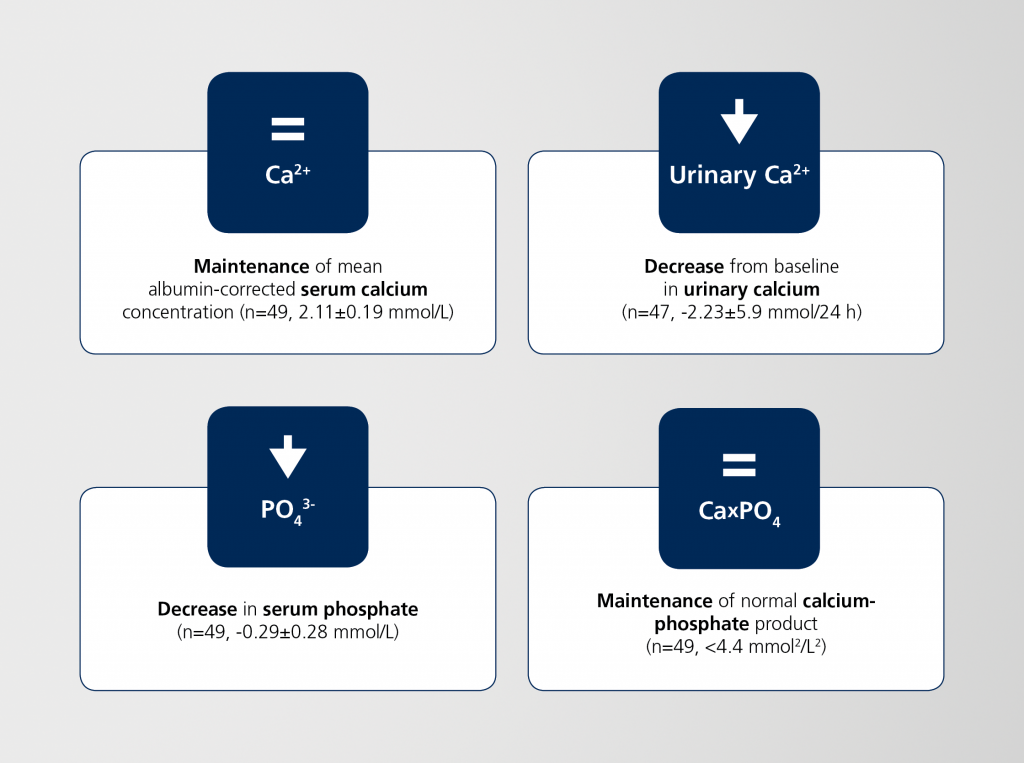
NATPAR has maintained efficacy for the long-term treatment of chronic hypoparathyroidism1,3
RACE is an open-label extension study to evaluate the long-term safety and efficacy of NATPAR.
A total of 49 patients have been enrolled in the study.1 The primary endpoint to assess the
long-term safety of NATPAR using flexible dosing (25, 50, 75, or 100 μg/day) was met; selected secondary efficacy outcomes are presented below.1,3
Current RACE results demonstrate durability of the physiological effects of NATPAR over 60 months, including:1
References
- NATPAR Summary of Product Characteristics.
- Mannstadt M, Clarke BL, Vokes T, et al. Efficacy and safety of recombinant human parathyroid hormone (1-84) in hypoparathyroidism (REPLACE): a double-blind, placebo-controlled, randomised, phase 3 study. Lancet Diabetes Endocrinol. 2013;1(4):275-83.
- Mannstadt M, Clarke BL, Bilezikian JP, et al. Safety and Efficacy of 5 Years of Treatment With Recombinant Human Parathyroid Hormone in Adults With Hypoparathyroidism. J Clin Endocrinol Metab. 2019;104(11):5136-47.
Natpar® (parathyroid hormone (rDNA)
Detailed Safety Information
Please consult the Natpar Summary Product Characteristics (SmPC) before prescribing.
Natpar treatment should be supervised by a physician or other qualified healthcare professional experienced in the management of patients with hypoparathyroidism. The goal of treatment is to achieve calcaemic control and to reduce symptoms. The optimisation of parameters of calcium phosphate metabolism should be in line with current therapeutic guidelines for the treatment of hypoparathyroidism. Prior to initiating and during treatment with Natpar confirm that 25-OH vitamin D stores are sufficient and that serum magnesium is within the reference range.
Contraindications
Natpar is contraindicated in patients with hypersensitivity to the active substance or to any of the excipients, who are receiving or who have previously received radiation therapy to the skeleton, with skeletal malignancies or bone metastases, who are at increased baseline risk for osteosarcoma, with unexplained elevations of bone-specific alkaline phosphatase, with pseudohypoparathyroidism.
Warnings and Precautions
Monitoring of patients during treatment: pre-dose and in some cases post-dose serum calcium levels must be monitored during treatment with Natpar.
Hypercalcaemia: this was reported in clinical trials with Natpar. Hypercalcaemia commonly occurred during the titration period, during which doses of oral calcium, active vitamin D, and Natpar were being adjusted. Hypercalcaemia may be minimized by following the recommended dosing, the monitoring information and asking patients about any symptoms of hypercalcaemia. If severe hypercalcaemia develops, hydration and temporarily stopping Natpar, calcium and active vitamin D should be considered until serum calcium returns to the normal range. Then consider resuming Natpar, calcium and active vitamin D at lower doses.
Hypocalcaemia: a common clinical manifestation of hypoparathyroidism was reported in clinical trials with Natpar. Most of the hypocalcaemic events occurring in the clinical trials were mild to moderate severity. The risk for serious hypocalcaemia was greatest after the withdrawal of Natpar. Temporary or permanent discontinuation of Natpar must be accompanied by monitoring of serum calcium levels and increase of exogenous calcium and/or vitamin D sources as necessary. Hypocalcaemia may be minimized by following the recommended dosing, the monitoring information, and asking patients about any symptoms of hypocalcaemia.
Concomitant use with cardiac glycosides: Hypercalcaemia of any cause may predispose to digitalis toxicity, monitor serum calcium and cardiac glycoside levels and patients for signs and symptoms of digitalis toxicity.
Severe renal or hepatic disease: Natpar should be used with caution in patients with severe renal or hepatic disease because they have not been evaluated in clinical trials.
Use in young adults: Natpar should be used with caution in young adult patients with open epiphyses.
Tachyphylaxis: the calcium-raising effect of Natpar may diminish over time in some patients. The response of serum calcium concentration to administration of Natpar should be monitored at intervals to detect this and the diagnosis of tachyphylaxis considered.
Urolithiasis: Natpar has not been studied in patients with urolithiasis. Natpar should be used with caution in patients with active or recent urolithiasis because of the potential to exacerbate this condition.
Adverse Reactions
The most commonly observed adverse events with Natpar treatment were hypercalcaemia, hypocalcaemia, headache, diarrhoea, vomiting, paraesthesia, hypoaesthesia and hypercalciuria.
|
Very common
(frequency ≥1/10): |
Hypercalcaemia, hypocalcaemia, headache, hypoaesthesia, paraesthesia, diarrhoea, nausea, vomiting, arthralgia, and muscle spasms. |
|
Common
(≥1/100 to <1/10): |
Hypomagnesaemia, tetany, anxiety, insomnia, somnolence, palpitations, hypertension, cough, upper abdominal pain, muscle twitching, musculoskeletal pain, myalgia, neck pain, pain in extremities, hypercalciuria, pollakiuria, asthenia, chest pain, fatigue, injection site reactions, thirst, anti-PTH antibody positive, blood 25-hydroxycholecalciferol decreased, vitamin D decreased. |
Material code: pi-00658
Date of Preparation of SI: February 2019
Prescribing Information
| Body weight (kg) | Volume to be injected (mL) |
|---|---|
| 38–41 | 0.xx |
| 42–45 | 0.xx |
| 46–49 | 0.xx |
| 50–53 | 0.xx |
| 54–57 | 0.xx |
| 58–61 | 0.xx |
| 62–65 | 0.xx |
| 66–69 | 0.xx |
| 70–73 | 0.xx |
| 74–77 | 0.xx |
| 78–81 | 0.xx |
| 82–85 | 0.xx |
| 86–89 | 0.xx |
| 90–93 | 0.xx |